 
from the University of Sydney two years later. Rutherford began his secondary education at a teachers’ training college in Auckland in 1889, then moved to Sydney to study commencing in 1890 and graduated with a B.A. #Rutherford experiment conclusion professional#When his father died at sea four years later, Rutherford became the ward of his uncle, a professional land surveyor. Rutherford was born on August 30, 1871, in Brightwater, New Zealand. Who is Ernest Rutherford Ernest Rutherford Life & Education This ionizing radiation can be a dangerous health hazard if present at high levels, but it is also the mechanism by which some medical imaging devices operate. In 1903, he became the first person to demonstrate that radioactive materials emit energy in a continuous stream of particles and are not merely energy sources. The radiation was focused into a narrow beam after passing through a slit in a lead screen.Ernest Rutherford Atomic Theory Model & Experiment Ernest Rutherford ContributionĮrnest Rutherford was a New Zealand-born British chemist and physicist known for his pioneering work in the study of radioactivity. #Rutherford experiment conclusion series#The nucleus was postulated as small and dense to account for the scattering of alpha particles from thin gold foil, as observed in a series of experiments performed by undergraduate Ernest Marsden under the direction of Rutherford and German physicist Hans Geiger in 1909.Ī radioactive source emitting alpha particles (i.e., positively charged particles, identical to the helium atom nucleus and 7,000 times more massive than electrons) was enclosed within a protective lead shield. 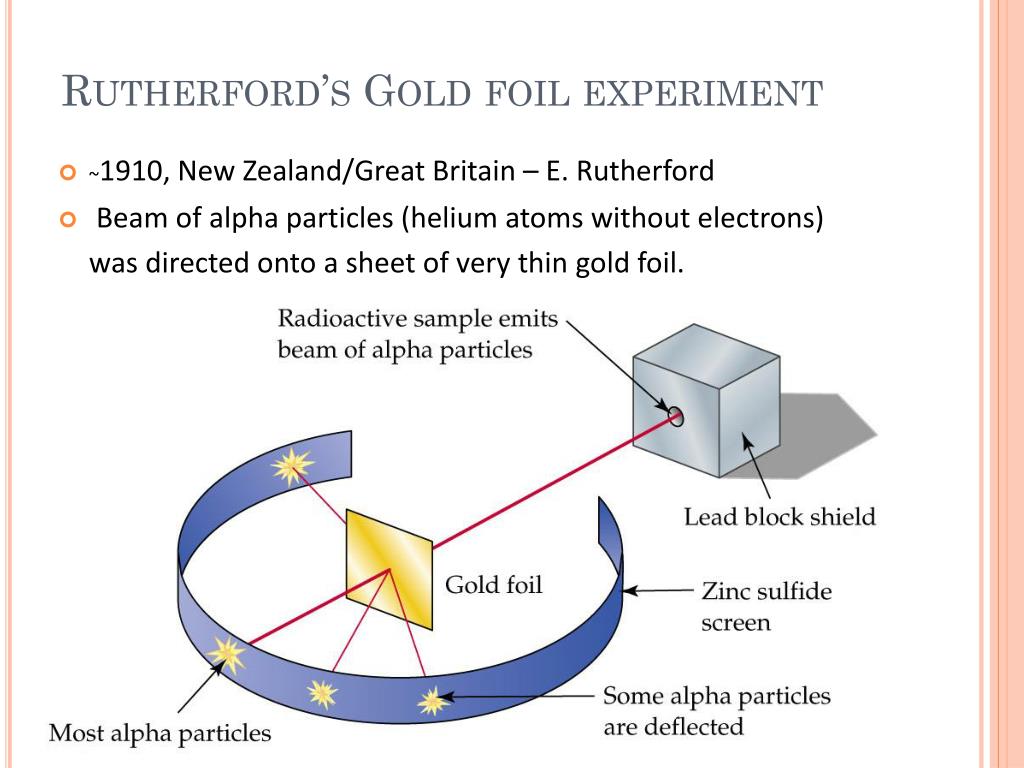
Rutherford Experiment Conclusion Series Of Experiments Niels Bohr Read more about Danish physicist Niels Bohr, the first physicist to apply quantum theory to the problem of atomic structure. In Bohrs model the orbits of the electrons were explained by quantum mechanics. What was the impact of Ernest Rutherfords theory The gold-foil experiment showed that the atom consists of a small, massive, positively charged nucleus with the negatively charged electrons being at a great distance from the centre. Quantum Read about the theory used in the Bohr model to more correctly describe the structure of atoms. In the Bohr model, which used quantum theory, the electrons exist only in specific orbits and can move between these orbits.Įlectromagnetic radiation Learn more about electromagnetic radiation and why it posed a problem for Ernest Rutherfords atomic model. This meant that an electron circling the nucleus would give off electromagnetic radiation. 
Most of the mass is in the nucleus, and the nucleus is positively charged.īut the Rutherford atomic model used classical physics and not quantum mechanics. What did Ernest Rutherfords atomic model get right and wrong The Rutherford atomic model was correct in that the atom is mostly empty space. Thomson atomic model Read about the Thomson atomic modelthe earliest model of atomic structureand how Ernest Rutherfords gold-foil experiment disproved it.īohr model Learn about Niels Bohrs atomic model and how it differs from Ernest Rutherfords atomic model. The Bohr atomic model, relying on quantum mechanics, built upon the Rutherford model to explain the orbits of electrons. What were the results of Rutherfords experiment The previous model of the atom, the Thomson atomic model, or the plum pudding model, in which negatively charged electrons were like the plums in the atoms positively charged pudding, was disproved. 
The only way this would happen was if the atom had a small, heavy region of positive charge inside it.Īlpha particle Learn about the particles used in the Rutherford gold-foil experiment. What is the Rutherford gold-foil experiment A piece of gold foil was hit with alpha particles, which have a positive charge. The neutron had not been discovered when Rutherford proposed his model, which had a nucleus consisting only of protons.Įrnest Rutherford Read more about Ernest Rutherford, the physicist whose gold-foil experiment revealed key information about the structure of atoms. #Rutherford experiment conclusion manual#
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
